This is not my first blog about salt- in 2013 I detailed the problems we are facing in the Northeast with rising salt levels in our fresh waters in this blog. I have continued the monitoring in Eagleville Brook that I described in the past blog, and now four years later, I have the ability to now look back at large dataset to get an idea of how salt levels have changed in one local stream.
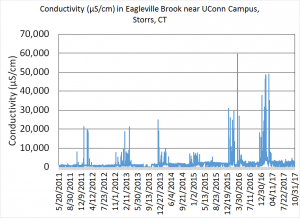
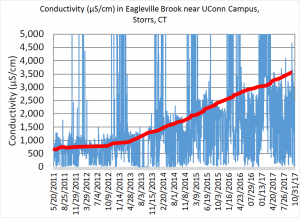
In Eagleville Brook, we have a long-term (6 years) record of discharge, temperature, conductivity and turbidity measurements (real-time data can be viewed here). Conductivity is a measure of how salty the water is; the higher the number, the more salt is in the water. Since 2011, we can see extremely high peaks of conductivity during the winter months when salty water runs off the impervious surfaces on campus (Figure 1). These high peaks are fairly short-lived, and are the result of melt water entering the stormwater system. The bigger concern is the conductivity in the stream during the summer months, when temperatures are higher, and flows are at their lowest. Salt that entered the ground surface (running off from sidewalks where there is not stormwater collection) during the winter slowly makes its way through soil and enters Eagleville Brook. The conductivity during these low flow periods has tripled since 2011. The level of conductivity in this stream is much higher than background levels, and is stressful for aquatic life.
Fortunately, UConn is taking action to reduce the amount of salt wasted during the winter months. Facilities staff are being trained by UConn’s technology transfer center on how to more efficiently apply deicing products while still providing safe travel for pedestrians and vehicles.

The goal of this program is to reduce excess salt applications, which has the double benefit of saving money and reducing the environmental impact. Facilities and environmental compliance staff at UConn are invested in making the program a success. I will report on progress next year!